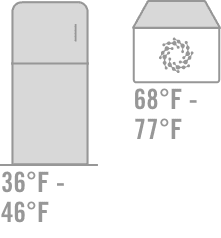
Approved Use
Repatha® is an injectable prescription medicine used:
- in adults with cardiovascular disease to reduce the risk of heart attack, stroke, and certain types of heart surgery. READ MORE
- along with diet alone or together with other cholesterol-lowering medicines in adults with high blood cholesterol levels called primary hyperlipidemia (including a type of high cholesterol called heterozygous familial hypercholesterolemia [HeFH]) to reduce low-density lipoprotein (LDL) or bad cholesterol.
For adults with heart disease